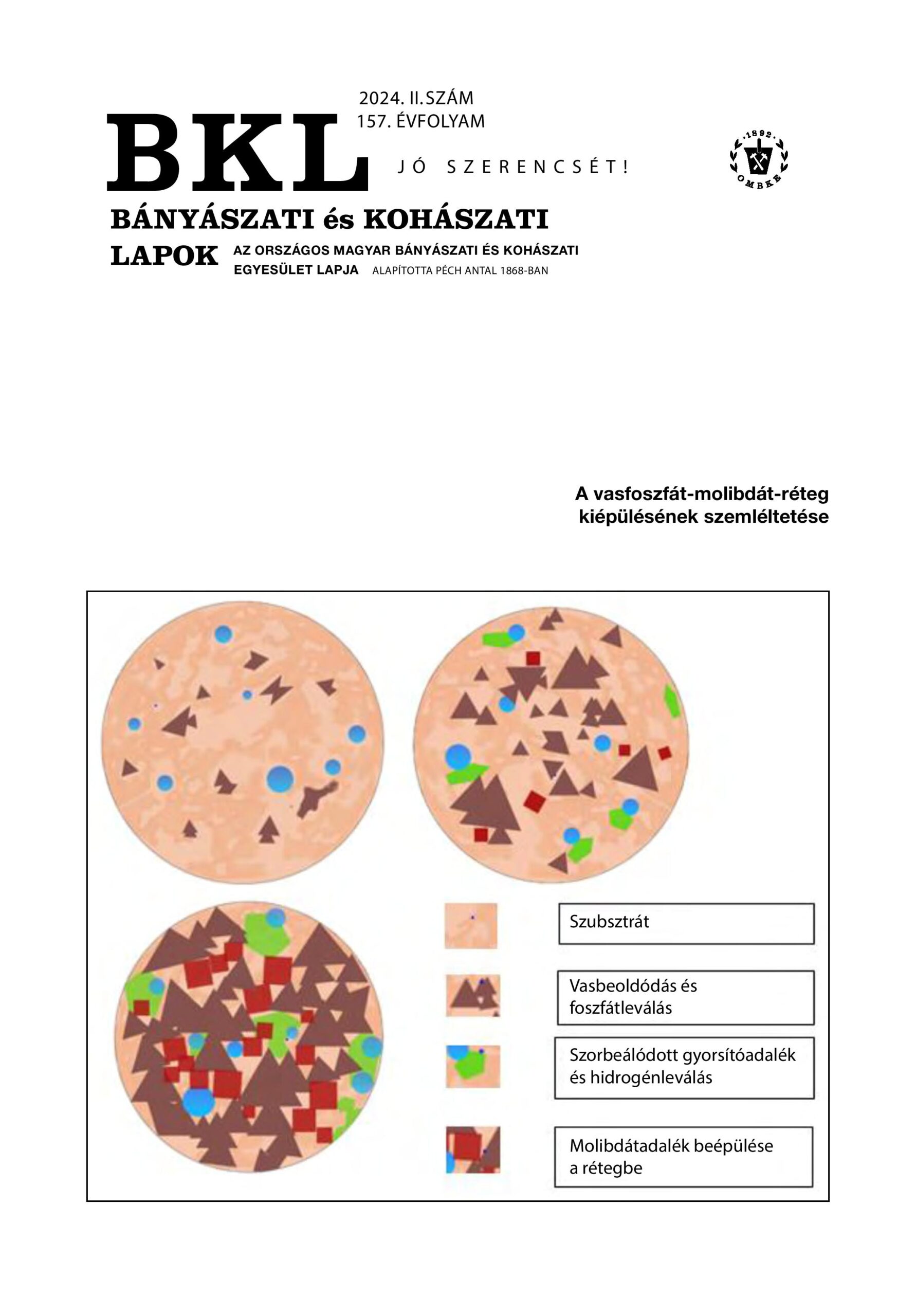
Evolution of phosphate-containing conversion layers in surface treatment: iron phosphating with molybdate additive
Abstract
In industrial phosphating processes, before cataphoretic dip/immersion painting, treatment solutions containing zinc phosphate are usually used to deposit the conversion thin interlayers in order to help adhere the paint coating better to the metal object. For the same purpose, iron phosphate layers are also developed on the surface of iron-base alloys (steels, cast irons), mostly by spraying and using appropriate additives in the treatment solutions. The zinc-phosphated surfaces are typically covered with a thin crystalline layer, whose chemical composition can usually be determined relatively easily, for example, by X-ray diff raction techniques. However, in the case of iron phosphating technology, only some kind of partially crystalline and only very thin conversion layers are formed on steel samples in the course of a longer-time dipping process, and mostly only under laboratory conditions. Industrial iron phosphate coatings are usually around 0.1 μm thick, amorphous in nature. Determining their exact chemical composition is a particularly diffi cult task. Nevertheless, during our own laboratory tests, still some valuable chemical element composition data could be obtained on our amorphous thin phosphate layers by performing qualitative analysis in depths profi le analysis via the argon plasma ablation technique with a glow discharge optical emission spectrometer (GD OES). During the GD OES sputtering, it was possible to detect important changes in the signal intensities of the Fe, P, Mo, Mg, Na, Ca, and K elements, although except the much less detectable oxygen and hydrogen, which, otherwise are the major bound elements of the formed “mixed” metals(Fe,Mg,Na,Ca,K,…)-phosphates-molibdates-oxide-hydroxide-bound-water type, rather complex thin surface conversion layers. Considering our numerous other test results (specifi c layer/deposit masses, optical microscopic (OM) and scanning electron microscopic (SEM) images, EDS X-ray analysis), it can be concluded that the iron phosphating process with molybdate additive can be a promising environmentally friendly and cost-saving alternative to the current industrial phosphating surface treatment processes developed so far and used in many surface fi nishing industries.
References
Yakubovich O., Khasanova N., Antipov E.: Mineral-inspired materials: Synthetic phosphate analogous for battery applications. Minerals, 10 (2020) 524. https://doi.org/10.3390/min10060524
Frost R. L., Weier M. L., Martens W., Kloprogge J. T., Ding Z.: Dehydration of synthetic and natural vivianite. Thermochimica Acta, 401(2) (2003) 121-130. https://doi.org/10.1016/S0040-6031(02)00505-1
Herbáth B., Kovács K., Jakab M., Makó É.: Crystal structure and properties of zinc phosphate layers on aluminum and steel alloy surfaces. Crystals, 13 (2023) 369. https://doi.org/10.3390/cryst13030369
al-Swaidani A. F.: Modified zinc phosphate coatings: a promising approach to enhance the anti-corrosion properties of reinforcing steel. MOJ Civil Engineering, 3(5) (2017) 370-374. https://doi.org/10.15406/mojce.2017.03.00083
Nickel and its compounds are included in REACH Annex XVII restricted substances list (entry 27). http://www.chemsafetypro.com/Topics/Restriction/reach_annex_XVII_nickle_compounds.html
Nagy T. Z.: Cink-foszfát helyettesítése nehézfémmentes előkezeléssel kataforetikus festősorokon. BKL Kohászat, 155(1), 2022, 17-19.
Popić J. P., et al.: The effect of deposition temperature on the surface coverage and morphology of iron-phosphate coatings on low carbon steel. Applied Surface Science, 257 (2011) 10855-10862. https://doi.org/10.1016/j.apsusc.2011.07.122
Popić J. P., Jegdić B. V., Bajat J. B., Mitrić M., Mišković-Stanković V. B.: Determination of surface coverage of iron-phosphate coatings on steel using the voltammetric anodic dissolution technique. J. Serb. Chem. Soc., 78(1) (2013) 101-114. https://doi.org/10.2298/JSC120706096P
Chen C., Liu G. B., Wang Y., Li J. L., Liu H.: Preparation and electrochemical properties of LiFePO4/C nanocomposite using FePO4∙2H2O nanoparticles by introduction of Fe3(PO4)2∙8H2O at low cost. Electrochimica Acta, 113 (2013) 464-469. https://doi.org/10.1016/j.electacta.2013.09.095
Jiang C., Gao Z., Pan H., Cheng X.: The initiation and formation of a double-layer phosphate conversion coating on steel. Electrochemistry Communications 114 (2020) 106676. https://doi.org/10.1016/j.elecom.2020.106676
Erdey-Grúz Tibor: Elektródfolyamatok kinetikája. Akadémiai Kiadó, 1969
Despić A. R., Dražić D. M., Savić-Maglić G. A., Atanasoski R. T.: Hydrogen evolution on pyrolytic graphite. Croatica Chemica Acta, 44 (1972) 79-87.
Miyata Y., Kuwahara Y., Asakura S., Shinohara T., Yakou T., Shiimoto K.: Hydrogen evolution reaction on spheroidal graphite cast iron with different graphite areas in sulfuric acid solutions. Electrochemistry, 82(2) (2014) 104-108. https://doi.org/10.5796/electrochemistry.82.104
Bockris J. O'M., Dražić D. M., Despić A. R.: Electrochim. Acta, 4 (1961) 325. https://doi.org/10.1016/0013-4686(61)80026-1
Mathew V., Kim S., Kang J., Gim J., Song J., Baboo J. P., Park W., Ahn D., Han J., Gu L., Wang Y., Hu Y-S., Sun Y-K., Kim J.: Amorphous iron phosphate: potential host for various charge carrier ions. NPG Asia Materials, 6, (2014) e138. https://doi.org/10.1038/am.2014.98
Verbrungen H., Baert K., Terryn H., De Graeve I.: Molybdate-phosphate conversion coatings to protect steel in a simulated concrete pore solution. Surface & Coatings Technology, 361 (2019) 280-291. https://doi.org/10.1016/j.surfcoat.2018.09.056